Prascend® (pergolide mesylate) has been available to U.S. veterinarians and horse owners for nearly three years as a treatment option for horses suffering from Pituitary Pars Intermedia Dysfunction (PPID), formerly known as equine Cushing’s disease. In an effort to raise awareness of the disease and share treatment success stories, Boehringer Ingelheim Vetmedica, Inc. (BIVI), manufacturers of PRASCEND, recently hosted a testimonial contest.
“The contest was designed to help us not only capture PRASCEND treatment success stories but also increase horse-owner awareness of the disease and its early clinical signs,” says Dwana Neal, brand manager, equine pharmaceuticals, BIVI. “Since PRASCEND was launched, we have received a number of success stories anecdotally, but through the contest we were not only able to draw attention to the disease, but also get a real sense of the impact PRASCEND has made for horses and owners dealing with this debilitating disease.”
The Barn Buzz Testimonial Contest was targeted to owners of horses diagnosed with PPID that had successful treatment results with PRASCEND. Horse owners visited a website and submitted their success stories for a chance to win prizes. One grand prize winner and three runners-up were selected from the entries. Winners were chosen based on the evaluation of written history, along with before and after photos.
BIVI is pleased to announce the following winners:
Grand Prize:
Skotch
Owned by Toni Bessler, Pertersburg, Ky.
Treated by Dr. Donna Mather, Donna Mather DVM

Runners-up:
Frodo
Owned by Joyce Dickson, Columbia Station, Ohio
Treated by Dr. Endie Peckham, Visiting Vet, Inc.

Margalo
Owned by Dena Sanders, Montrose, Colo.
Treated by Dr. Becky Sondergard, Uncompahgre Equine Veterinary Services

Walker
Owned by Christy Whitelaw, Christianburg, Va.
Treated by Dr. Charlotte Hatch, Appalachian Veterinary Services
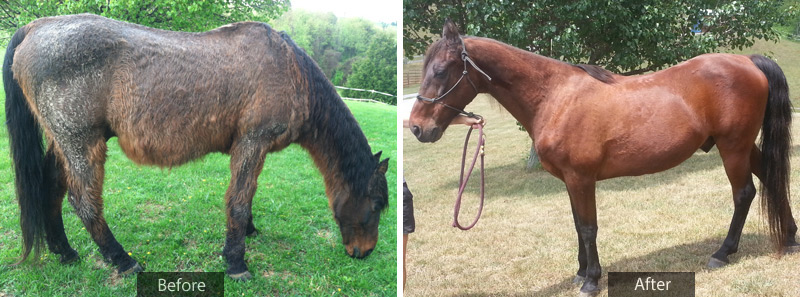
“It was difficult selecting a grand prize winner,” Neal adds, “but Skotch’s story was truly compelling and his improvement after starting treatment was quite dramatic.”
Skotch’s owner, Toni Bessler, noticed this spring that Skotch wasn’t shedding out the excessive amount of long, wavy hair he had acquired. “He was sweating a lot and breathing hard this summer and spent a lot of time in the barn trying to cool off. He was also losing muscle mass and looked thin and depressed,” says Bessler.
She contacted her veterinarian, Donna Mather, DVM, who performed the recommended adrenocorticotropic hormone (ATCH) test on Skotch which showed his ACTH level to be 321pg/mL, far above the normal range.* Mather and Bessler decided that PRASCEND would be the best treatment option for Skotch. Knowing in some horses that the initiation of treatment with PRASCEND may cause a transient decrease in appetite, lack of energy and possible change in behavior, Skotch was monitored closely by Dr. Mathers over the following three 3 weeks.
PRASCEND comes in tablet form. To avoid crushing of the tablet and increase risk of human exposure, it may be dissolved in water, sweetener or flavoring, and administered orally with a syringe.
Bessler saw improvement quickly after starting treatment. “Within three weeks, he was starting to shed his winter coat,” Bessler said. “He was also starting to pick up some weight and even looked happier. I saw him running in the field more and moving a little quicker than he used to. Within two months, he had lost almost all of his long winter hair and was beginning to look slick and shiny. He doesn’t sweat any more than the other horses do on our hot Kentucky summer days, and his muscle condition improved tremendously,” Bessler added.
“Oftentimes, subtle changes can be the earliest signs of the disease,” says Steve Grubbs, DVM, PhD, DACVIM, equine technical manager, BIVI. “Many horses will demonstrate early clinical signs of the disease but owners aren’t always aware these are PPID indicators, such as decreased athletic performance, lethargy and delayed shedding.”
Grubbs adds that horse owners should continue to work with their veterinarian after treatment has started. “Finding the right dosage and continuing to monitor the horses once they have begun treatment is essential to continued success,” says Grubbs.
PRASCEND is the first and only FDA-approved drug for treatment of PPID in horses. If you think your horse may have PPID, talk to your veterinarian. To read the complete Barn Buzz Testimonial Contest success stories, visit www.prascend.com.
Important safety information: PRASCEND is for use in horses only. PRASCEND has not been evaluated in breeding, pregnant or lactating horses and may interfere with reproductive hormones in these horses.
References:
*For more information on ACTH testing, visit the Equine Endocrinology Group website at https://sites.tufts.edu/equineendogroup/.
PRASCEND is a registered trademark of Boehringer Ingelheim Vetmedica GmbH, licensed to Boehringer Ingelheim Vetmedica, Inc. ©2014 Boehringer Ingelheim Vetmedica, Inc.
About Boehringer Ingelheim Vetmedica, Inc.
Boehringer Ingelheim Vetmedica, Inc., (BIVI) is a leader in the animal health industry with its portfolio of innovative vaccine and pharmaceutical solutions for the prevention and treatment of disease within the swine, cattle, equine and pet markets. BIVI is the U.S. division of the global Boehringer Ingelheim Animal Health GmbH. Our U.S. headquarters are located within the KC Animal Health Corridor in St. Joseph, Mo., with other sites in Ames, Fort Dodge and Sioux Center, Iowa.
The Boehringer Ingelheim group is one of the world’s 20 leading pharmaceutical companies. Headquartered in Ingelheim, Germany, it operates globally with 142 affiliates and more than 47,400 employees. Since it was founded in 1885, the family-owned company has been committed to researching, developing, manufacturing and marketing new medications of high therapeutic value for human and veterinary medicine.
Social responsibility is a central element of Boehringer Ingelheim’s culture. Involvement in social projects, caring for the employees and their families and providing equal opportunities for all employees form the foundation of the global operations. Mutual cooperation and respect, as well as environmental protection and sustainability are intrinsic factors of all Boehringer Ingelheim’s endeavors.
With nearly 3,000 employees worldwide, Boehringer Ingelheim Animal Health achieved net sales of about $1.46 billion (1.07 billion euros) in 2013. In our research-driven Animal Health business, Boehringer Ingelheim continually invests more than 10% of net sales of the Animal Health business in R&D.
For more information please visit www.bi-vetmedica.com





